 In fact, many proteins have a mixture of a helices, b sheets, and other types of folding patterns to form various overall shapes. A single polypeptide chain may have different regions that take on different secondary structures. These include various loops, helices and irregular conformations. While the a helix and b sheet are by far the most common types of structure, many others are possible. The major constituent of silk (silk fibroin) consists mainly of layers of b sheet stacked on top of each another. In this arrangement, side chains project alternately upward and downward from the sheet. Generally, the primary structure folds back on itself in either a parallel or antiparallel arrangement, producing a parallel or antiparallel b sheet. Again, the polypeptide N-H and C=O groups form hydrogen bonds to stabilize the structure, but unlike the a-helix, these bonds are formed between neighbouring polypeptide (b) strands. In a b sheet, the polypeptide chain folds back on itself so that polypeptide strands like side by side, and are held together by hydrogen bonds, forming a very rigid structure. The structure of a b sheet is very different from the structure of an a-helix. An example of a protein with many a-helical structures is the keratin that makes up human hair. The side chains project outward and contact any solvent, producing a structure something like a bottle brush or a round hair brush. The alpha helix has precise dimensions: 3.6 residues per turn, 0.54 nm per turn. All C=O and N-H groups are involved in hydrogen bonds, making a fairly rigid cylinder. Each carbonyl is linked by a hydrogen bond to the N-H of a residue located 4 residues further on in the sequence within the same chain. In this conformation, the carbonyl and N-H groups are oriented parallel to the axis. 
Various types of secondary structures have been discovered, but by far the most common is the orderly repeating forms known as the a-helix and the b sheet.Īn a helix, as the name implies, is a helical arrangement of a single polypeptide chain, like a coiled spring. This level of structure describes the local folding pattern of the polypeptide backbone and is stabilized by hydrogen bonds between N-H and C=O groups. Since the average molecular weight of a residue is about 110 Da, that single-chain has a molecular weight of over 3 million Daltons. There is no theoretical maximum size, but the largest protein so far discovered has about 30,000 residues. Even such small proteins contain hundreds of atoms and have molecular weights of over 5000 Daltons (Da). So the primary structure of a small protein would consist of a sequence of 50 or so residues. The minimum size of a protein is defined as about 50 residues smaller chains are referred to simply as peptides. Thus primary structure involves only the covalent bonds linking residues together. Primary structure is simply the sequence of residues making up the protein. It is convenient to discuss protein structure in terms of four levels (primary to quaternary) of increasing complexity. This constrains the flexibility of the chain and prevents some folding patterns. This means that the six atoms involved are coplanar, and that there is no free rotation around the C–N axis. The peptide bond is written as a single bond, but it actually has some characteristics of a double bond because of the resonance between the C-O and C-N bonds: If stretched out, the side chains of the individual residues project outwards from this backbone. This is referred to as the “backbone” of the peptide. 
The connection gives rise to a repeating pattern of “NCC-NCC-NCC…” atoms along the length of the molecule. Conventionally, peptides are written N-terminal first therefore gly-ser is not the same as ser-gly or GS is not the same as SG. In the natural course of making a protein, polypeptides are elongated by the addition of amino acids to the C-terminal end of the growing chain. One end has a free amino group (called the “N-terminal”) and the other has a free carboxyl group (“C-terminal”). 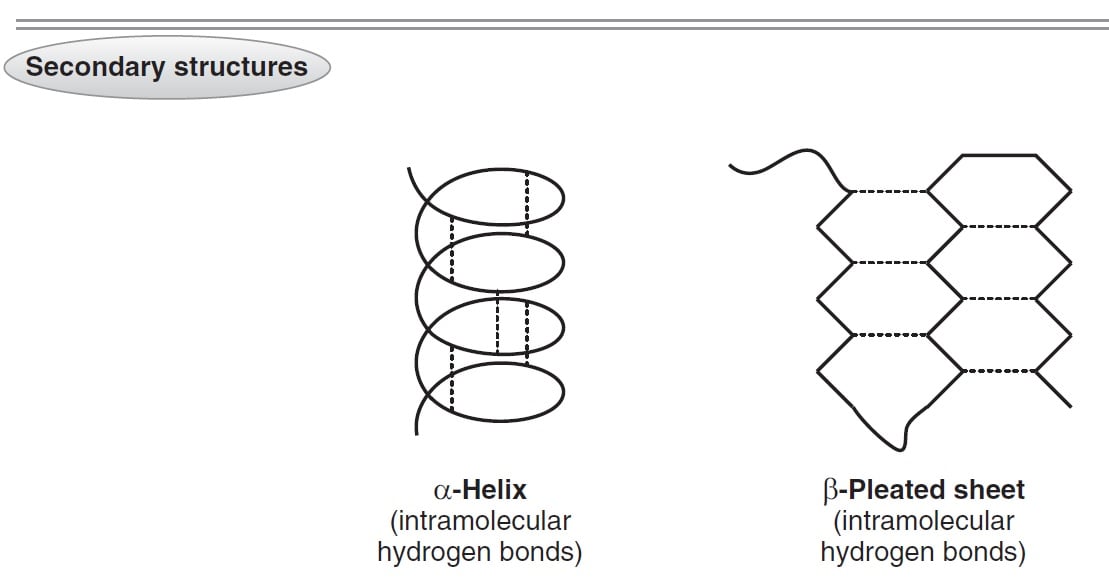
Another property of peptides is polarity: the two ends are different. Multiple amino acids result in a polypeptide (often shortened to “peptide”).īecause water is lost in the course of creating the peptide bond, individual amino acids are referred to as “amino acid residues” once they are incorporated. When two amino acids join, the result is called a dipeptide, three gives a tripeptide, etc. 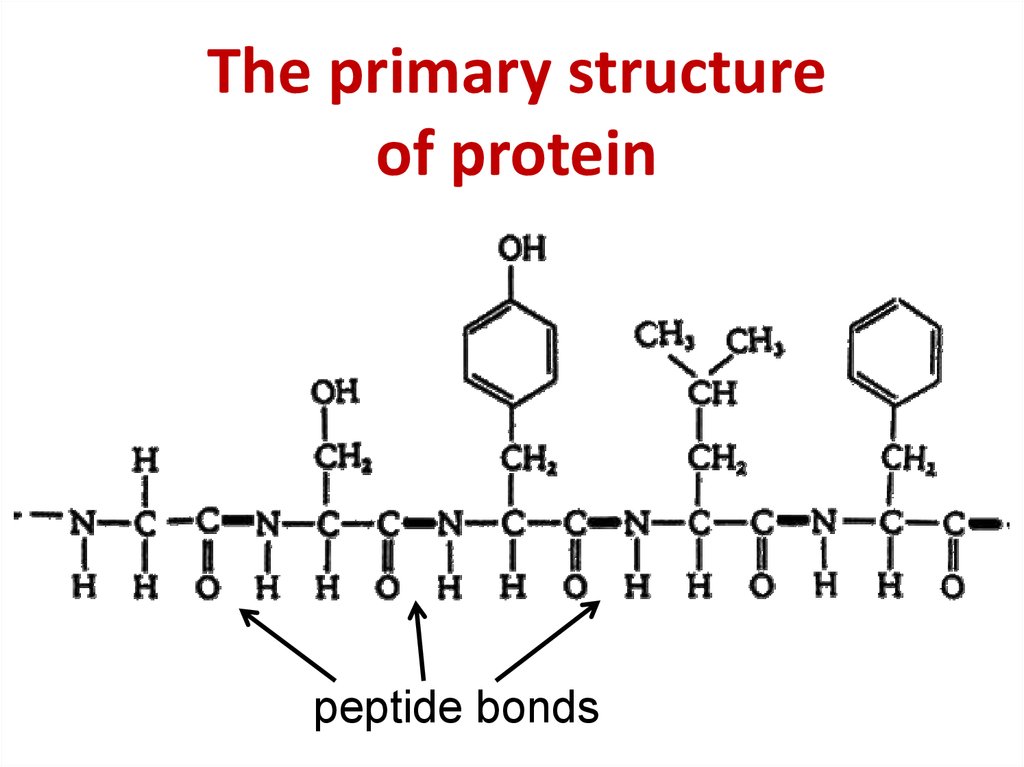
This bond is formed between the alpha amino group of one amino acid and the carboxyl group of another in a condensation reaction. To make a protein, amino acids are connected together by a type of amide bond called a “peptide bond”. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
